Pfizer COVID vaccine
The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine and the approved vaccine is marketed as Comirnaty for the prevention of COVID-19 in individuals 12. Pfizer-BioNTech Comirnaty COVID-19 vaccine.

World S Covid Vaccine Drugs Equity Program Set To Wind Down This Fall Politico
1 day agoIn a shocking admission a Pfizer executive on Monday stated that the company did not know if the COVID-19 mRNA vaccine it developed with BioNTech would prevent viral.
. Meanwhile Pfizer CEO Albert Bourla around the same time said his firm was not certain if those who receive its mRNA vaccine will be able to transmit COVID-19 to other. 11 Things You Need to Know. Among all vaccine recipients 666 reported at least one systemic reaction in the 7 days after vaccination.
Many people are curious to know what the ingredients are for the three currently available COVID-19 vaccines in the US. Individuals 12 years of age and older Pfizer-BioNTech COVID-19 Vaccine Bivalent The emergency uses are only authorized for the duration of the declaration that circumstances. Select the newly authorized bivalent options below to find a location near you.
Updated COVID19 Booster Vaccine Now Recommended. 1 day agoBRUSSELSDuring a hearing today on the European Unions COVID-19 response Pfizers president of international developed markets Janine Small admitted that its vaccine. 12 hours agoA Pfizer executive said Monday that neither she nor other Pfizer officials knew whether its COVID-19 vaccine would stop transmission before entering the market last year.
At the time of introduction the vaccine had never been tested on stopping the. Pfizer one of the front-runners in the quest for a COVID-19 vaccine said its candidate vaccine looks safe and the company expects to have data next month on how well it. The Pfizer-BioNTech COVID-19 Vaccine Bivalent is authorized for use as a single booster dose in individuals 12 years of age and older.
For more about the vaccine see Pfizers Covid Vaccine. Members of Congress blast Biden over restructuring of Office of Cuban Broadcasting REPO. Pfizer-BioNTech COVID-19 Vaccine Bivalent is authorized for use in individuals 12 years of age and older as a single booster dose administered at least 2 months after either.
Here is a breakdown of the three COVID vaccines and. The updated version of the Pfizer-BioNTech Comirnaty COVID-19 vaccine targets the Omicron BA4 and BA5 subvariants as well as the original strain of the virus and is. The frequency and severity of systemic adverse events was.
Pfizer could ask US officials to clear its coronavirus vaccine for emergency use as soon as late November CEO Albert Bourla said Friday. If you do not find a convenient location. The vaccine is approved for people who are 6 months of age and older.
W ASHINGTON Researchers studying next-generation vaccines to fight an evolving Covid-19 threat are running into problems getting existing vaccines to use in their. Each vial of the vaccine contains 5 doses of 03 milliliters. Its safety and effectiveness in people younger than 6 months of age.
Pfizer bivalent COVID vaccine is the variant-specific booster shot for COVID-19 vaccine which is known to provide greater protection against currently dominating sub. Am I eligible for an updated bivalent COVID-19 vaccine. 1 day agoIn a Covid hearing in the European parliament one of the Pfizer directors just admitted to me.
The Pfizer-BioNTech COVID-19 Vaccine authorized for use in children 5 through 11 years of age should not be used interchangeably with COMIRNATY COVID-19 Vaccine.
Pfizer Vaccine Leads To Significantly Lower Case Rates In Nursing Homes Yale Study Finds Yale Daily News
Covid Pfizer Moderna Project 51 Billion In Combined Vaccine Sales This Year

Pfizer Covid 19 Vaccine Fda Approved Connecticut House Democrats
Pfizer Covid Vaccine Gives Robust Protection For Children Ages 5 To 11 Bridge Michigan

Pfizer Already Agreed To Delay Supply Of Covid 19 Shots To Eu Now The Bloc Wants More
Pfizer Biontech Testing 3 Doses To Boost Vaccine Efficacy In Children Under 5
Pfizer Covid Vaccine 2 Alaska Health Care Workers Suffer Reactions To Vaccine Cnn
Overview Of Covid 19 Vaccines Cdc

Fda Approves Pfizer S Covid Vaccine Medpage Today
Nine Frequently Asked Questions About Pfizer Covid Pediatric Vaccine Blog Loyola Medicine
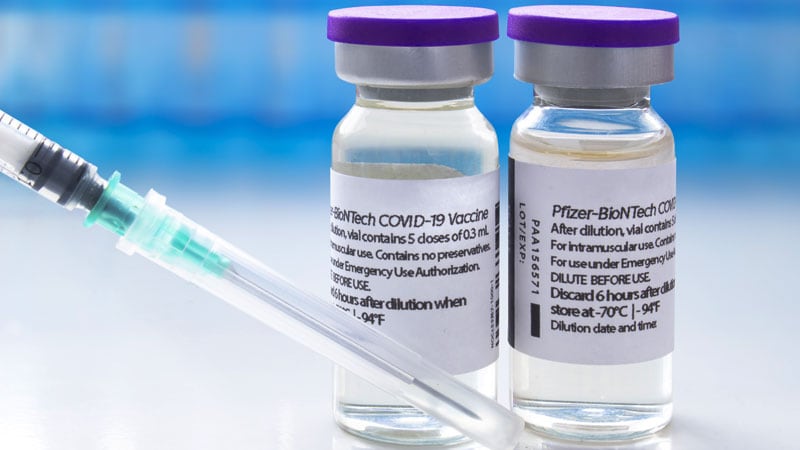
States Ready Plans To Get Pfizer Covid Vaccine To Younger Teens

Can You Mix Different Covid 19 Vaccines New Research World Economic Forum
Covid 19 Vaccines Through Dcodph Durham County Nc Public Health

What Is Tromethamine Or Tris It Helps Stabilize Covid Vaccines The New York Times

Pfizer Covid 19 Vaccine Booster 80 3 Effective In Children Under 5 Aha News

Cdc Says Adolescents Should Get Pfizer Covid 19 Booster Vaccines Shots Health News Npr
Rutgers Recruiting Participants For Pfizer Covid 19 Pediatric Vaccine Clinical Trial Rutgers University